
Laparoscopic radical antegrade modular pancreatosplenectomy for pancreatic cancer: technical tips and pitfalls
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is an aggressive disease with a poor prognosis. Although surgical resection is the only curative treatment, the 5-year survival rate after pancreatectomy for PDAC is approximately 20% (1). The major recurrence pattern after pancreatectomy is distant metastasis. However, local recurrence has also been reported to be high (2). Obtaining negative margins in surgical resection is key to reducing local recurrences.
Radical antegrade modular pancreatosplenectomy (RAMPS) is one of the operative methods for distal pancreatectomy, which was first reported in 2003 (3). It has been reported to be useful for obtaining a negative retro-pancreatic margin (4,5), possibly reducing local recurrence (6). With advances in minimally invasive surgery, laparoscopic RAMPS (L-RAMPS) has been performed for PDAC in the body and tail of the pancreas. Several studies have shown comparable short- and long-term outcomes between open RAMPS and L-RAMPS (7-9). The advantages of L-RAMPS include the magnified view of laparoscopy possibly with three-dimensional imaging system, which enables an understanding of the precise plane for dissection during surgery. However, at the same time, L-RAMPS for PDAC is technically demanding, and a lack of experience can lead to serious intraoperative and postoperative complications.
There are two types of L-RAMPS according to the extent of posterior dissection: anterior and posterior L-RAMPS. The difference between these operations is whether to resect the left adrenal gland. We performed anterior L-RAMPS, in which posterior dissection proceeded in front of the adrenal gland, for PDAC without invasion of the adrenal gland. A review of the literature was performed and our standard L-RAMPS technique was described.
Indications and preoperative evaluation
The indications for L-RAMPS can vary depending on the skill and experience of the surgeon. In our institution, we consider L-RAMPS for PDAC in the body and tail of the pancreas with neither direct invasion to the adjacent organs (except for the left adrenal gland) nor abutment to the major vessels, such as the celiac axis (CeA), superior mesenteric artery (SMA), and portal or superior mesenteric vein. Although invasion of the splenic vessels is not a contraindication, a distance of less than 5 mm between the tumor and the origin of the splenic artery (SpA) should be a contraindication for L-RAMPS as a safe ligation and division of the SpA is difficult. The distance between the tumor and the gastroduodenal artery is critical if a tumor extends into the neck of the pancreas. Laparoscopic dissection along the gastroduodenal artery is technically difficult and often requires the division of the superior pancreaticoduodenal artery. Injuries to the adventitia of the gastroduodenal artery can cause arterial bleeding after surgery.
Regarding anatomical factors, the location and tortuosity of the SpA are important in deciding how to approach it. The location of the origin of the dorsal pancreatic artery should be carefully checked in each case (10). It can easily be injured during dissection around the common hepatic artery (CHA) and SpA. If the dorsal pancreatic artery is abnormally dilated, it possibly supplies collateral flow into the liver from the SMA due to stenosis of the CeA (11). In such cases, perfusion of the hepatic artery should be carefully checked after division of the dorsal pancreatic artery. It should be checked preoperatively whether the arterial arcade in the head of the pancreas also supplies hepatic arterial flow. The presence of a portal annular pancreas is also important since it requires additional pancreatic transection behind the portal vein (12). The junction of the left gastric and inferior mesenteric veins should be checked to decide whether to preserve them or not. The anatomy of the left renal artery (LRA) should be checked as there may be an accessory renal artery or branching variations (13). The checklist for preoperative evaluation is shown in Table 1. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this article and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Table 1
| Tumor factors |
| Abutment of the major vessels* |
| Invasion of the adjacent organs |
| Distance to the root of the SpA |
| Distance to the gastroduodenal artery |
| Anatomical factors |
| Location of the SpA |
| Origin of the dorsal pancreatic artery |
| Location of the LRA |
| Junction of the left gastric/inferior mesenteric vein |
| Anatomical anomaly |
| CeA stenosis |
| Portal annular pancreas |
*, the CeA, SMA, and portal/superior mesenteric vein. SpA, splenic artery; LRA, left renal artery; CeA, celiac axis; SMA, superior mesenteric artery.
Surgical techniques
Trocar placement
The patient was placed in the reverse Trendelenburg position, with the legs spread. Five trocars were placed in the upper abdomen. In most procedures, the operator is on the right side and the assistant is on the left side of the patient (Figure 1A). However, the positions were switched during some procedures (Figure 1B-1D).

Retraction of the liver and stomach
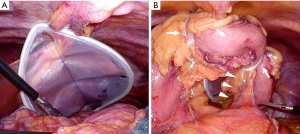
For adequate lymphadenectomy, liver retraction is necessary to obtain a wide operative field around the superior margin of the pancreas. Several instruments, including the Nathanson liver retractor, have been reported to be useful for liver retraction (14-16). We used a Silicon DiskTM (Hakko Co., Ltd., Nagano, Japan) to retract the liver (Figure 2A). 2-0 Prolene® (Ethicon, Somerville, NJ, USA) was used to anchor it to the diaphragm around the esophageal hiatus and the abdominal wall below the xiphoid process. This method is less invasive than using a Nathanson liver retractor because only 2-0 Prolene® passes through the abdominal wall. In addition to retraction of the liver, retraction of the stomach is also key to a wide exposure of the pancreas (17-20). We used two 6-cm Penrose drains to retract the stomach. After incisions of the lesser and greater omentum, the left gastroepiploic and short gastric vessels were transected. The two Penrose drains passed behind the stomach and were pulled into the antrum and body of the stomach (Figure 2B). This method can provide adequate tension to the left gastric vessels, making lymphadenectomy easy to perform.
Ligation of the SpA
It is important to first identify and ligate the SpA to reduce bleeding around the spleen and prevent pancreato-splenic congestion after ligation of the splenic vein. Although some reports have shown the efficacy of the artery-first approach, in which the SpA is ligated and divided before pancreatic transection (21-23), we usually only ligate and do not divide the SpA first, since it is often difficult to expose the root of the SpA widely before pancreatic transection.
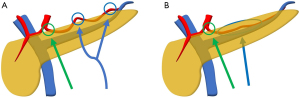
There are two options to approach SpA: superior and inferior approaches (Figure 3). In most cases, the superior approach, with dissection around the superior margin of the pancreas on the left side of the CHA, can expose the SpA near its origin. However, in some cases, it is difficult to identify the SpA near its origin due to the tumor location or inflammation around the pancreas. In such cases, identification of the SpA at a distal location is a possible option. As the SpA usually runs tortuously along the superior margin of the pancreas (24), there are some points where it can be easily identified by the superior approach (Figure 3A). However, for cases in which the SpA runs relatively straight behind the pancreas, the inferior approach is useful (Figure 3B) (25). A proper approach should be chosen based on the tumor location and anatomy of the SpA in each case.
Exposure of the left renal vein (LRV)
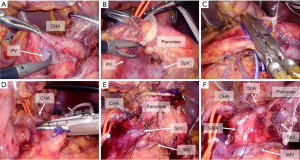
The LRV is a useful landmark to secure the retropancreatic margin of the tumor. There are two different approaches to the LRV: (I) approach from the cranial side of the transverse mesocolon (Figure 4A) (13,26,27); and (II) approach from the caudal side of the transverse mesocolon (the ligament of Treitz approach, Figure 4B) (27-29). We routinely perform the ligament of Treitz approach for two reasons. First, the laparoscopic caudal view makes it easier to approach the caudal side of the transverse mesocolon. Second, it has the advantage of securing a negative retro-pancreatic margin, especially when the tumor is close to the caudal aspect of the pancreas. An intraoperative image of the ligament of Treitz approach is shown in Figure 4C. We closed a defect in the transverse mesocolon after the ligament of Treitz approach because it can cause postoperative ileus (30).

Figure 4D shows three types of posterior dissection according to the tumor location. Some landmarks determine the margins of the posterior dissection. The medial margin should be the SMA because it makes it easy to determine the margin of lymphadenectomy for No. 14 (lymph nodes along the SMA). The left adrenal gland is a useful landmark for determining the cranial margin of dissection. The lateral margin can vary depending on the tumor location, and dissection should be extended to the left margin of the tumor. For tumors close to the left kidney, lateral dissection should be extended to the left, along the anterior surface of the kidney. In such cases, surgeons should be careful about LRA since it sometimes runs above the LRV near the renal hilum (13). If invasion of the adrenal gland is suspected, posterior dissection should proceed behind the adrenal gland (posterior L-RAMPS).
Transection of the pancreas and splenic vessels
Before transecting the pancreas at the neck, we routinely encircle the CHA. During this procedure, preservation of the nerve plexus of the CHA is important for preventing arterial injury (Figure 5A). Invasion of the nerve plexus of the CHA is very rare when the tumor does not abut it in preoperative computed tomography (CT) images (31). Next, the plane between the pancreatic neck and the portal or superior mesenteric vein should be widely dissected (Figure 5B). A polyester surgical loop was passed behind the pancreatic neck and used for retraction during pancreatic transection. There are some tips on stapler pancreatic transection to prevent postoperative pancreatic fistula: (I) precompression of the pancreas (32-35); (II) adequate selection of the cartridge (36-38); (III) slow firing technique (39). We used two parallel intestinal clamps for five minutes of precompression of the pancreas (Figure 5C). A cartridge with a length of 45 mm and a height of 1.5–2.25 mm is adequate in most cases (Figure 5D), but it should be changed according to the width, thickness, and hardness of the pancreas. A firing speed of 30 s was applied every 1 cm.
After transection of the pancreas, the root of the splenic vein was widely exposed (Figure 5E). They were dissected and divided using double ligation. Retraction of the pancreatic body to the left exposed the root of the SpA (Figure 5F). It is important to dissect the nerve plexus around the SpA at the location of the ligation to prevent slipping of the ligatures. It also creates an appropriate distance for the ligation and division of the SpA. Double ligation is usually performed for the SpA with 3-0 Vicryl® (Ethicon, Somerville, NJ, USA) and a Hem-o-lok® clip (Weck Closure Systems, Research Triangle Park, NC, USA).
En bloc lymphadenectomy
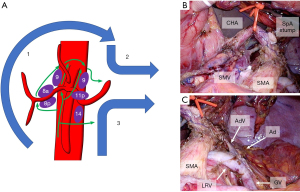
In the tumor, node, metastasis (TNM) classification, the regional lymph nodes for PDAC in the body and tail of the pancreas are the lymph nodes along the CHA, CeA, SpA, and splenic hilum, as well as the retroperitoneal nodes and lateral aortic nodes (40). However, the extent of lymphadenectomy for PDAC in the body and tail of the pancreas is controversial. While the International Study Group of Pancreatic Surgery recommended lymphadenectomy for only Nos. 9 (lymph nodes around the CeA), 10 (lymph nodes at the splenic hilum), 11p (lymph nodes along the proximal SpA), 11d (lymph nodes along the distal SpA), and 18 (lymph nodes along the inferior margin of the pancreas) for distal pancreatectomy (41), some studies have demonstrated nodal involvement in Nos. 8a (lymph nodes in the anterosuperior group along the CHA), 8p (lymph nodes in the posterior group along the CHA), and 14 (lymph nodes along the SMA), especially in PDAC in the body of the pancreas (42,43). Our standard lymphadenectomy includes removal of Nos. 8a, 8p, 9, 10, 11p, 11d, 14, and 18 lymph nodes. Figure 6A shows the order of dissection in our en bloc lymphadenectomies. Lymph nodes 8a and 8p were dissected along the CHA. Then, the No. 9 (right side) lymph nodes were continuously dissected along the crus and the CeA. The dissected lymph nodes were pulled to the left through the back of the left gastric vessels. The Nos. 9 (left side) and 11p lymph nodes were dissected along the CeA and SpA. Finally, the No. 14 lymph nodes were dissected along with the SMA. The dissected plane of No. 14 lymph nodes should be continuous with the plane of the exposed LRV. Sampling of the No. 16a2 lat (lateral para-aortic lymph nodes between the upper margin of the CeA and the lower margin of the LRV) was performed for staging. Intraoperative images after lymphadenectomy are shown in Figure 6B,6C.
Dissection of the retroperitoneal tissue and retrieval of the specimen
After en bloc lymphadenectomy, dissection of the retroperitoneal tissue was performed along the body and tail of the pancreas and spleen. A retrieval bag was placed in the left subphrenic space. The specimen was bagged in a “spleen-first” fashion and retrieved via the umbilicus with a minimal port site incision. The cut margin of the pancreas was routinely assessed using frozen section analysis. A peritoneal drain was routinely placed on the stump of the pancreas.
Conclusions
Several important anatomical structures can be recognized preoperatively for L-RAMPS. Surgeons should be familiar with the multiple approaches to landmark vessels during L-RAMPS. Given the variations in anatomy and location of the tumor, selecting the proper approach for each case leads to safety and oncological benefits.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Ippei Matsumoto) for the series “Laparoscopic Pancreatic Surgery” published in Laparoscopic Surgery. The article has undergone external peer review.
Peer Review File: Available at https://ls.amegroups.com/article/view/10.21037/ls-22-46/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://ls.amegroups.com/article/view/10.21037/ls-22-46/coif). The series “Laparoscopic Pancreatic Surgery” was commissioned by the editorial office without any funding or sponsorship. The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this article and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Oba A, Croce C, Hosokawa P, et al. Prognosis Based Definition of Resectability in Pancreatic Cancer: A Road Map to New Guidelines. Ann Surg 2022;275:175-81. [Crossref] [PubMed]
- Groot VP, Rezaee N, Wu W, et al. Patterns, Timing, and Predictors of Recurrence Following Pancreatectomy for Pancreatic Ductal Adenocarcinoma. Ann Surg 2018;267:936-45. [Crossref] [PubMed]
- Strasberg SM, Drebin JA, Linehan D. Radical antegrade modular pancreatosplenectomy. Surgery 2003;133:521-7. [Crossref] [PubMed]
- Abe T, Ohuchida K, Miyasaka Y, et al. Comparison of Surgical Outcomes Between Radical Antegrade Modular Pancreatosplenectomy (RAMPS) and Standard Retrograde Pancreatosplenectomy (SPRS) for Left-Sided Pancreatic Cancer. World J Surg 2016;40:2267-75. [Crossref] [PubMed]
- Grossman JG, Fields RC, Hawkins WG, et al. Single institution results of radical antegrade modular pancreatosplenectomy for adenocarcinoma of the body and tail of pancreas in 78 patients. J Hepatobiliary Pancreat Sci 2016;23:432-41. [Crossref] [PubMed]
- Kiritani S, Kaneko J, Arita J, et al. Radical antegrade modular pancreatosplenectomy for left-sided pancreatic ductal adenocarcinoma may reduce the local recurrence rate. Dig Surg 2022; [Crossref]
- Kawabata Y, Hayashi H, Kaji S, et al. Laparoscopic versus open radical antegrade modular pancreatosplenectomy with artery-first approach in pancreatic cancer. Langenbecks Arch Surg 2020;405:647-56. [Crossref] [PubMed]
- Huang J, Xiong C, Sheng Y, et al. Laparoscopic versus open radical antegrade modular pancreatosplenectomy for pancreatic cancer: a single-institution comparative study. Gland Surg 2021;10:1057-66. [Crossref] [PubMed]
- Hirashita T, Iwashita Y, Fujinaga A, et al. Surgical and oncological outcomes of laparoscopic versus open radical antegrade modular pancreatosplenectomy for pancreatic ductal adenocarcinoma. Surg Today 2022;52:224-30. [Crossref] [PubMed]
- Horiguchi A, Ishihara S, Ito M, et al. Multislice CT study of pancreatic head arterial dominance. J Hepatobiliary Pancreat Surg 2008;15:322-6. [Crossref] [PubMed]
- Heo S, Kim HJ, Kim B, et al. Clinical impact of collateral circulation in patients with median arcuate ligament syndrome. Diagn Interv Radiol 2018;24:181-6. [Crossref] [PubMed]
- Ohtsuka T, Mori Y, Ishigami K, et al. Clinical significance of circumportal pancreas, a rare congenital anomaly, in pancreatectomy. Am J Surg 2017;214:267-72. [Crossref] [PubMed]
- Nishino H, Nagakawa Y, Takishita C, et al. Safe exposure of the left renal vein during laparoscopic distal pancreatectomy for pancreatic ductal adenocarcinoma: anatomical variations and pitfalls. Surg Today 2020;50:1664-71. [Crossref] [PubMed]
- Shibao K, Higure A, Yamaguchi K. Disk suspension method: a novel and safe technique for the retraction of the liver during laparoscopic surgery (with video). Surg Endosc 2011;25:2733-7. [Crossref] [PubMed]
- Shinohara T, Kanaya S, Yoshimura F, et al. A protective technique for retraction of the liver during laparoscopic gastrectomy for gastric adenocarcinoma: using a Penrose drain. J Gastrointest Surg 2011;15:1043-8. [Crossref] [PubMed]
- Kitajima T, Shinohara H, Haruta S, et al. Prevention of transient liver damage after laparoscopic gastrectomy via modification of the liver retraction technique using the Nathanson liver retractor. Asian J Endosc Surg 2015;8:413-8. [Crossref] [PubMed]
- Surjan RC, Basseres T, Makdissi FF, et al. Innovative technique for gastric retraction during laparoscopic distal pancreatectomy: the marionette. J Surg Case Rep 2015;2015:rjv157. [Crossref] [PubMed]
- Morikawa T, Ishida M, Takadate T, et al. The superior approach with the stomach roll-up technique improves intraoperative outcomes and facilitates learning laparoscopic distal pancreatectomy: a comparative study between the superior and inferior approach. Surg Today 2020;50:153-62. [Crossref] [PubMed]
- Dokmak S, Aussilhou B, BenSafta Y, et al. Double Gastric Hanging for Gastric Exposure in Laparoscopic Distal Pancreatectomy. Dig Surg 2019;36:449-54. [Crossref] [PubMed]
- Hirono S, Hayata K, Kawai M, et al. Complete REtraction of the StomaCh using pEnrose draiN and liver reTractor (CRESCENT) during laparoscopic distal pancreatectomy. Langenbecks Arch Surg 2020;405:1243-50. [Crossref] [PubMed]
- Takaori K, Uemoto S. Artery-First Distal Pancreatectomy. Dig Surg 2016;33:314-9. [Crossref] [PubMed]
- Tian F, Sun MQ, Lu J, et al. Retrograde artery first approach for "shoulder" pancreatic cancers in minimally invasive distal pancreatectomy. Surg Endosc 2021;35:74-80. [Crossref] [PubMed]
- Nagai K, Kiguchi G, Yogo A, et al. Left-posterior approach for artery-first en bloc resection in laparoscopic distal pancreatectomy for left-sided pancreatic cancer. Langenbecks Arch Surg 2020;405:1251-8. [Crossref] [PubMed]
- Sylvester PA, Stewart R, Ellis H. Tortuosity of the human splenic artery. Clin Anat 1995;8:214-8. [Crossref] [PubMed]
- Inoko K, Ebihara Y, Sakamoto K, et al. Strategic Approach to the Splenic Artery in Laparoscopic Spleen-preserving Distal Pancreatectomy. Surg Laparosc Endosc Percutan Tech 2015;25:e122-5. [Crossref] [PubMed]
- Yamamoto M, Zaima M, Yamamoto H, et al. New laparoscopic procedure for left-sided pancreatic cancer-artery-first approach laparoscopic RAMPS using 3D technique. World J Surg Oncol 2017;15:213. [Crossref] [PubMed]
- Kato T, Inoue Y, Oba A, et al. Laparoscopic Radical Antegrade Modular Pancreatosplenectomy with Anterocranial Splenic Artery-First Approach for Left-Sided Resectable Pancreatic Cancer (with Videos). Ann Surg Oncol 2022;29:3505-14. [Crossref] [PubMed]
- Sunagawa H, Harumatsu T, Kinjo S, et al. Ligament of Treitz approach in laparoscopic modified radical antegrade modular pancreatosplenectomy: report of three cases. Asian J Endosc Surg 2014;7:172-4. [Crossref] [PubMed]
- Ome Y, Hashida K, Yokota M, et al. Laparoscopic radical antegrade modular pancreatosplenectomy for left-sided pancreatic cancer using the ligament of Treitz approach. Surg Endosc 2017;31:4836-7. [Crossref] [PubMed]
- Nanno Y, Goto T, Toyama H, et al. Internal hernia through a transverse mesocolon defect after laparoscopic distal pancreatectomy: Report of a case. Asian J Endosc Surg 2017;10:187-90. [Crossref] [PubMed]
- Okada K, Uemura K, Kondo N, et al. Prognostic significance of dissecting the nerve plexus around the common hepatic artery in pancreatic cancer. Langenbecks Arch Surg 2021;406:679-89. [Crossref] [PubMed]
- Nakamura M, Ueda J, Kohno H, et al. Prolonged peri-firing compression with a linear stapler prevents pancreatic fistula in laparoscopic distal pancreatectomy. Surg Endosc 2011;25:867-71. [Crossref] [PubMed]
- Ariyarathenam AV, Bunting D, Aroori S. Laparoscopic Distal Pancreatectomy Using the Modified Prolonged Prefiring Compression Technique Reduces Pancreatic Fistula. J Laparoendosc Adv Surg Tech A 2015;25:821-5. [Crossref] [PubMed]
- Hirashita T, Ohta M, Yada K, et al. Effect of pre-firing compression on the prevention of pancreatic fistula in distal pancreatectomy. Am J Surg 2018;216:506-10. [Crossref] [PubMed]
- Asbun HJ, Van Hilst J, Tsamalaidze L, et al. Technique and audited outcomes of laparoscopic distal pancreatectomy combining the clockwise approach, progressive stepwise compression technique, and staple line reinforcement. Surg Endosc 2020;34:231-9. [Crossref] [PubMed]
- Miyasaka Y, Mori Y, Nakata K, et al. Attempts to prevent postoperative pancreatic fistula after distal pancreatectomy. Surg Today 2017;47:416-24. [Crossref] [PubMed]
- Sugimoto M, Kendrick ML, Farnell MB, et al. Relationship between pancreatic thickness and staple height is relevant to the occurrence of pancreatic fistula after distal pancreatectomy. HPB (Oxford) 2020;22:398-404. [Crossref] [PubMed]
- Nishikawa M, Yamamoto J, Hoshikawa M, et al. Stapler sizes optimized for pancreatic thickness can reduce pancreatic fistula incidence after distal pancreatectomy. Surg Today 2020;50:623-31. [Crossref] [PubMed]
- Matsumoto I, Kamei K, Satoi S, et al. Efficacy of the slow firing method using a reinforced triple-row stapler for preventing postoperative pancreatic fistula during laparoscopic distal pancreatectomy. Surg Today 2022;52:260-7. [Crossref] [PubMed]
- Brierley JD, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours. 8th ed. Hoboken: John Wiley & Sons, 2017.
- Tol JA, Gouma DJ, Bassi C, et al. Definition of a standard lymphadenectomy in surgery for pancreatic ductal adenocarcinoma: a consensus statement by the International Study Group on Pancreatic Surgery (ISGPS). Surgery 2014;156:591-600. [Crossref] [PubMed]
- Imamura T, Yamamoto Y, Sugiura T, et al. Reconsidering the Optimal Regional Lymph Node Station According to Tumor Location for Pancreatic Cancer. Ann Surg Oncol 2021;28:1602-11. [Crossref] [PubMed]
- Ishida H, Ogura T, Takahashi A, et al. Optimal Region of Lymph Node Dissection in Distal Pancreatectomy for Left-Sided Pancreatic Cancer Based on Tumor Location. Ann Surg Oncol 2022;29:2414-24. [Crossref] [PubMed]
Cite this article as: Ishida J, Toyama H, Goto T, Fukumoto T. Laparoscopic radical antegrade modular pancreatosplenectomy for pancreatic cancer: technical tips and pitfalls. Laparosc Surg 2023;7:8.


